This post reflects on the Tobacco Free Generation (TFG) policy - a regulation to ban the sale of tobacco products to people born after a certain year.
(A
subsequent post by Stan Glantz with additional concerns can be read here.)
AMBIGUOUS: One term, two concepts
The term "tobacco-free generation" is applied two distinct concepts, both of which are used with varying scope.
Some use the terms to describe a public health objective: the point at which smoking or tobacco use is no longer picked up by young people as a result of strong conventional tobacco control policies. Examples of tobacco free generation being discussed in this way by researchers and advocates can be found in Europe (including conference resolutions) and the United States.
This post focuses on the term as it is used to describe a phased-in legislated ban on tobacco sales. An example can be found in a recent draft law for England and Wales: "(1) It is an offence to sell any of the following to a person born on or after 1 January 2009." The term "smoke-free generation" is synonymously used.
VAGUE: One term, variable scope
In some jurisdictions (i.e. the United States) tobacco is defined to include e-cigarettes, and TFG would (unless otherwise specified) apply also to e-cigarettes. The Brookline Bylaw, for example, takes this approach.
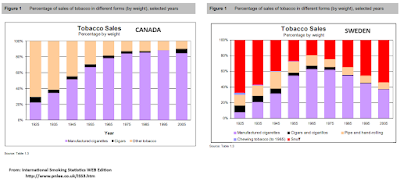
In other jurisdictions (i.e. Canadian federal law) the term "tobacco" applies strictly to products containing tobacco leaf. The U.K. and New Zealand proposals discussed below did not impose any restrictions on the sale of e-cigarettes or other modern nicotine products, but focused restrictions on traditional combustible tobacco (in both countries traditional oral tobacco has long been banned).
NASCENT: an untested proposition
The idea of phasing in a ban on tobacco sales was first proposed in 2010 and campaigned for by medical professionals based in Singapore. Since then the concept has been discussed around the world, with an increased level of interest in the past few years. No country has yet implemented it, although a small number of cities in the United States have.
The first legislature to consider TFG was the Australian state of Tasmania. Hearings on a proposed bill were held in 2015, but the measure died on the order paper.
In July 2021, the town of Brookline, Massachusetts received state approval for a by-law that banned the sale of cigarettes to anyone born after January 1, 2000. Brookline is a satellite city of Boston, and four neighbouring communities have also adopted this measure. A legal challenge which delayed the initial implementation of the law was resolved earlier this year when the bylaw was upheld by the state's upper court. It is now due to come into effect on January 1, 2025. These laws apply equally to tobacco and vaping products.
In December 2022, revisions to New Zealand's Smokefree Environments and Regulated Products Act were made which included a ban on the sale of tobacco products to anyone born after January 1, 2009. After a general election the following year and a change of government, the NZ legislature reversed these amendments and removed them from the law.
TFG had a second false start in a national legislature in 2024. In March 2024, the U.K. government introduced the concept as part of its Tobacco and Vapes Bill, received submissions from the public and debated the issue in Committee. The House of Commons had not finished its study of the bill before a general election was called on May 30. Similar to the New Zealand proposal, the U.K. government intended to ban the sale of tobacco products to people born after January 1, 2009 but not to impose such constraints on non-tobacco nicotine products (such as vaping or pouches).
The Hong Kong government included TFG measures in its 2023 consultation on future tobacco control initiatives. A year later, however, this was not included among the 10 measures the government chose to implement.
TOPICAL: under review in 2 Canadian jurisdictions
This spring two Canadian provinces have launched public consultations about TFG measures.
On May 14, 2024, Canada's smallest province issued a "Consultation Paper on Wellness in Prince Edward Island" which included TFG among 4 proposals to help the province "Breathe Well". "No one born after Jan. 1, 2009 would be able to legally purchase cigarettes. The effect of a TFG approach is to stop the next generation of youth from purchasing tobacco products."
Prince Edward Island already has in place Canada's most advanced restrictions to protect young people from nicotine use. Four years ago it increased the minimum age to buy tobacco or vaping products from 19 to 21 and to only permit the sale of vaping products in specialty stores. A year later it banned flavourings in e-cigarettes.
The other proposals included in this consultation are to permit tobacco to be sold only in tobacconist shops, to increase public education investments to prevent youth initiation of tobacco and vaping and to expand supports for smoking cessation. Public comments on these proposals are due by July 5, 2024, and a survey tool is available to PEI residents to respond.
On May 30, 2024, Newfoundland and Labrador announced that it would consult on changes to its tobacco law. The on-line consultation will be open until July 8, 2024. Our answers to the Newfoundland consultation are pasted at the end of this post.
NON-DISRUPTIVE: minimal impact in the short term
A recent article written by tobacco control leaders identified some benefits and risks of "birthdate-based commercial tobacco sales." (Malone RE, McAfee T. Birthdate-based commercial tobacco sales restrictions: will ‘tobacco-free generation’ policies advance or delay the endgame? Tobacco Control Published Online First: 19 June 2024. doi: 10.1136/tc-2024-058716).
They identify that such "have intuitive appeal, largely because they do not appreciably disrupt the status quo of retail sales, which continue unchanged for all those born before the designated cut-off date." This, combined with the minimal impact on short-term tax revenues, enhances their "political palatability"
These authors conclude that birthdate-based restrictions will not greatly accelerate reductions in tobacco use and does not satisfy the need for more profound change. "[Birthdate-based Sales Restriction] makes only
gradual changes to the structural, political and social dynamics
that sustain the epidemic, as the population to whom the products may be legally sold grows older and the use of the products
perhaps appears less appealing to young adults."
These authors - who were among the first to promote a tobacco Endgame - do not rate this as a sufficiently powerful public policy. "Overall, as an endgame policy, compared with other proposed
policies BSR is the temporally weakest and least disruptive to the
status quo, allowing the tobacco industry to continue promoting
lethal products, retailers to continue selling them and permitting
the tobacco epidemic to continue for decades as the eligible to-buy population gradually declines."
SUB-OPTIMAL: stronger new interventions are available
Better endgame measures are available, these authors suggest. Among the alternatives they propose are:
* A shorter phase-out of retail sales of tobacco [including vaping and modern oral] products (1–5
years), ideally combined with transition funding
and education for retailers as well as education and cessation
support for tobacco users....
* A rapid ban on sales of combustible tobacco products,
coupled with additional restrictions on non-combustible
sales but without an immediate ban...
* Major restrictions on retail outlet density, number and types
of stores...
* ..Bans on sales of flavoured products, allowing sales only
of products with nicotine at non-addictive levels and/
or marketing/advertising bans.
These American researchers based their analysis on U.S. circumstances, where tobacco is legally defined to include vaping and other nicotine products. Consequently, they did not profile the inherent problems of banning one form of nicotine sales while allowing another.
For governments like Newfoundland and Prince Edward Island, this is a crucial consideration. Banning sale of tobacco products to come individuals, while allowing the sale of other addictive and harmful products made by the same manufacturers sends a signal. Consumers will interpret that alternative nicotine products are not very harmful (or else they would be embraced by the restriction). The industry will encourage the distinction to be interpreted as an endorsement of its plans to recruit new generations to its newer products.
------------------------
Response of Physicians for a Smoke-Free Canada to Newfoundland's consultations on Amendments to Tobacco and Vaping-related Statutes
1. Should individuals born after a certain year (e.g., born after 2008) be prohibited from purchasing tobacco and related products?
Our answer is a conditional yes.
We recommend that such a prohibition should only be implemented if it is accompanied by a similar prohibition on the sale of vaping and other current or future recreational nicotine products.
Such a prohibition should not apply to nicotine used in a therapeutic context (e.g. supplied through therapeutic channels like pharmacies and used for the purpose of treating addiction or other disease).
The benefits of a tobacco free generation measure that does not include vaping products is likely to be small. Cigarette smoking is passé among newer generations, just as pipe smoking became passé among their grandparents. This means that a prohibition on tobacco use among people born after 2008 may not result in lower use of tobacco products than if no such measure were in place. For example, the Canadian Tobacco and Nicotine Survey showed that 8% of Canadians born between 1998 and 2003 smoked cigarettes, but 20% used vaping products.
The risks of a tobacco-free generation measure that does not include vaping products are significant. They include:
* young people perceiving that there is government approval for the use of these products after the age of 19 (or 21)
* the industry being able to point to government as a validator of its position that the widespread sale of vaping products should be tolerated or encouraged and that rules that apply to tobacco products should not apply to vaping, pouches or other nicotine 'starter' products.
The province should explore alternative ways to accelerate reductions in tobacco use, including mandatory phase-outs of combustible tobacco and other nicotine products (potentially in different time-frames), retail restrictions including tobacco- or nicotine-only stores, strict liability for tobacco or nicotine manufacturers when young people use their products, etc.
2. Should the legal age for the purchase of tobacco and related products be increased to 21?
This measure should be implemented as soon as possible and should apply to all products for which youth use is a concern.
3. Should non-tobacco flavoured vaping products, such as candy, fruit, and mint flavours, be prohibited?
Yes.
The failure of the federal government to fulfill its obligations by implementing flavour restrictions has unreasonably placed the burden of protecting youth on provincial governments. Newfoundland should adopt this measure while also calling for federal measures. Without a federal ban, provincial bans are easily undermined by cross-border internet sales.
A ban on flavourings should also include restrictions on the types of ingredients than can be used, including mint-menthol additives and sweeteners.
4. Should the sale of nicotine pouches be restricted, such as by only allowing sales at pharmacies?
Yes.
Nicotine pouches may (or may not) prove to be a much safer form of nicotine use than vaping, chewing tobacco or smoked tobacco. However, it now appears that the most likely users of these products are young people who are not currently smokers, including young adults who are being targeted with messages encouraging nicotine use as a way to improve concentration, mood or to reduce weight. Restricting sales to pharmacies is one way to ensure that they are used in ways which promote health, and do not serve as a on-ramp to nicotine addiction.
5. Should the sale of tobacco and related products through vending machines be prohibited?
Yes.
This measure is long overdue and should be implemented as soon as possible. On-line sale of tobacco and vaping products should also not be permitted, except in exceptional circumstances regarding remote communities.
6. Should vapour product retailers be licensed?
Yes.
The licensing requirements on vapour product retailers should include an obligation to report sales volumes to the province. This will permit the province to assess the impact of unrestricted number of retail outlets on nicotine use at a neighbourhood level.
7. Should there be stricter enforcement measures and/or penalties in legislation governing the sale of tobacco and related products?
Yes.
Tobacco and nicotine companies should also bear responsibility for the sale to and use by underage users. A suitably adapted strict liability system should be in place to penalize those who sell products in ways that facilitate youth use. For example, the websites of British American Tobacco (VUSE) and Philip Morris (VEEBA) offer volume discounts in on-line sales, which provides an economic incentive for social sales. Strict liability for underage use would encourage these companies to adopt measures to ensure no spillage of their supply into social or illicit sales.